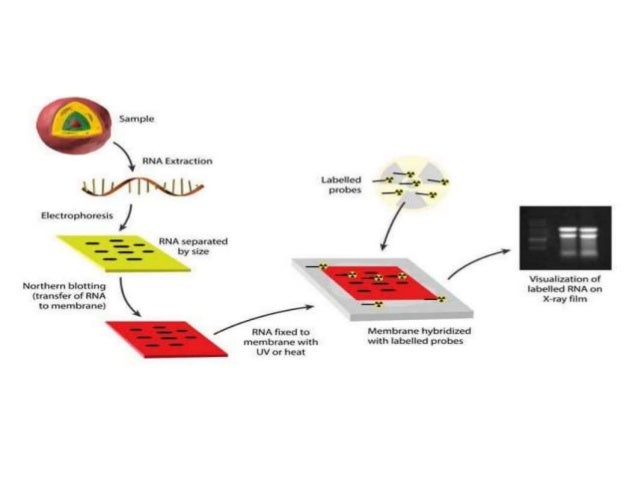
In this article, a commonly used protocol and expected results are described and discussed. The whole Southwestern blotting procedure takes ∼4 d to complete. This technique could be combined with database searching (TransFac, ), prediction of potential protein factors binding onto a target motif (e.g., Patch search), in vitro supershift EMSA and in vivo chromatin immunoprecipitation (ChIP) assays for effective identification of protein factors. The interaction of the probe with the protein(s) is later visualized by autoradiography. This method combines the features of Southern and Western blotting techniques a denaturing SDS-PAGE is first employed to separate proteins electrophoretically based on size, and after transferring the proteins to a membrane support, the membrane-bound proteins are renatured and incubated with a 32P-labeled double-stranded oligonucleotide probe of specific DNA sequence. The advantage of this technique over other related methods such as electrophoretic mobility shift assay (EMSA) and DNA footprinting is that it provides information regarding the molecular weight of unknown protein factor. Southwestern blotting is used to investigate DNA–protein interactions. The successful detection of transcription factor binding in a Southwestern blot is also a prerequisite for using DNA affinity chromatography and screening of an expression library with DNA probes in the subsequent purification and isolation of the transcription factor. For example, Southwestern blotting is used to estimate the molecular mass of a transcription factor. All of these methods are based on the specific binding of protein transcription factors to DNA, but they differ in their respective roles in the characterization of transcription factors. Further experiments identified the nuclear protein bound to this region as nuclear factor-1 (NF1) or a closely related protein by using Southwestern blotting, DNA affinity chromatography, UV crosslinking of proteins to DNA, methylation interference analysis, and DNA gel-mobility shift assays (4). Footprinting analyses using liver nuclear extracts have revealed a strongly protected region between −490 and −540 (oligo II) within the P2 promoter region (3). In order to clarify the molecular mechanisms of its transcription, we have cloned the rat α 1B-AR gene and identified its promoters and the cis-acting elements in its 5′ regulatory domain (2). Expression of the α 1B-AR gene is controlled by hormonal and developmental factors in a complex, tissue-specific manner. The α 1B-adrenergic receptor (α 1B-AR) is a G-protein-coupled receptor that plays a key role in the sympathetic regulation of a variety of physiological processes, such as cardiac and smooth muscle contractility, contraction of the spleen, liver glycogenolysis, melatonin secretion by the pineal gland, and cell proliferation and hypertrophy (1).
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
